Every year, U.S. hospitals hemorrhage between $28 billion and $45 billion treating preventable healthcare-associated infections—a staggering financial burden that premium nitrile glove selection can dramatically reduce. Behind these numbers lies a critical truth: the difference between standard and premium barrier protection directly impacts infection rates, litigation exposure, and regulatory compliance costs. Understanding Healthcare-Associated Infection Liability: How Premium Nitrile Glove Selection Reduces $28-45B Annual Costs requires examining the documented performance metrics that justify investment in superior hand protection.
Healthcare facilities face mounting pressure to demonstrate infection control effectiveness while managing tight budgets. The solution increasingly centers on evidence-based glove procurement strategies that prioritize tensile strength, pin-hole rates, and chemical resistance over lowest-bid purchasing. This approach transforms glove selection from a commodity decision into a strategic liability mitigation tool.
Key Takeaways
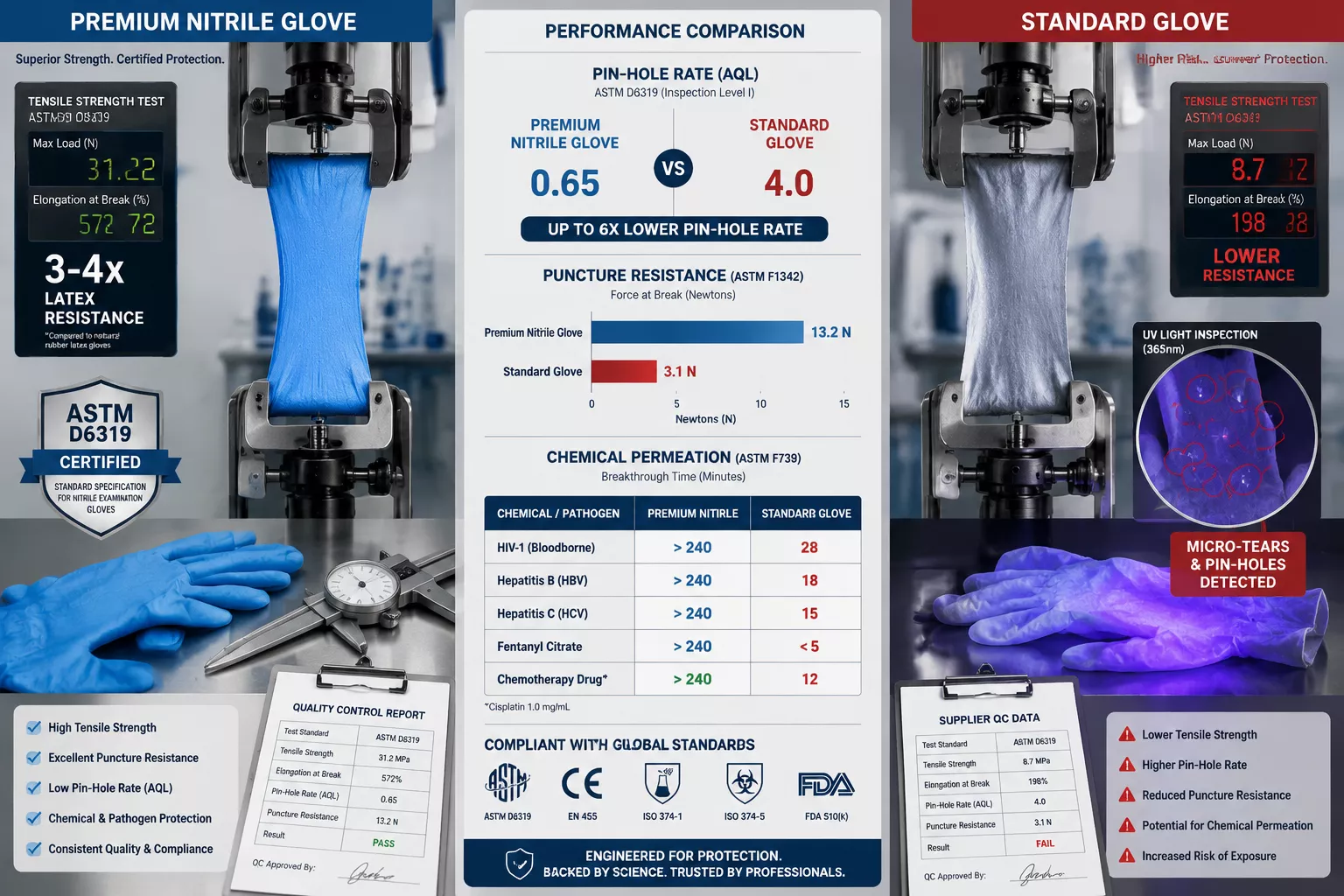
- Premium nitrile gloves reduce HAI risk by 30-70% through superior puncture resistance (3-4x stronger than latex) and barrier integrity
- Annual HAI costs of $28-45 billion can be significantly lowered by selecting medical-grade nitrile meeting ASTM D6319 and EN 455 standards
- Documented traceability and compliance records protect facilities from OSHA violations and infection-control litigation
- Thickness selection (6-8 mil for high-risk tasks) and AQL ratings directly correlate with reduced exposure incidents
- Proper training on donning/doffing maximizes the 30-70% HAI reduction potential of premium nitrile gloves
The Financial Case for Premium Nitrile: Understanding Healthcare-Associated Infection Liability Costs
Healthcare-associated infections impose massive direct and indirect costs on medical facilities. Direct treatment expenses include extended hospital stays, additional medications, and specialized care procedures. Indirect costs encompass litigation settlements, regulatory penalties, reputation damage, and lost revenue from reduced patient volumes.
Why premium nitrile matters financially:
- Puncture resistance: Nitrile exam gloves provide 3-4 times greater puncture resistance than latex alternatives, critical in emergency rooms and operating theaters where sharps exposure risks run highest
- Chemical barrier performance: Superior resistance to bloodborne pathogens, solvents, acids, and pharmaceutical compounds like fentanyl reduces exposure incidents that trigger workers’ compensation claims
- Latex allergy elimination: With 5-10% of healthcare workers experiencing latex sensitivity, powder-free nitrile prevents allergic reactions that compromise glove integrity and inflate liability costs
Studies published in the Journal of Hospital Infection demonstrate that correct use of premium nitrile gloves decreases HAI risk by up to 70%. The American Journal of Infection Control reports a 30% reduction in HAIs following nitrile adoption across hospital systems. These reductions translate directly to billions in avoided treatment costs and liability exposure.
Documented Performance Metrics That Justify Premium Investment
Hospitals increasingly justify premium nitrile investments through quantifiable performance data:
| Performance Metric | Premium Nitrile | Standard Latex | Impact on Liability |
|---|---|---|---|
| Puncture Resistance | 3-4x stronger | Baseline | Fewer breach incidents |
| Pin-Hole Rate (AQL) | 1.5 or lower | 2.5-4.0 | Reduced pathogen exposure |
| Chemical Permeation | Excellent (oils, solvents) | Poor | Lower chemical injuries |
| Tensile Strength | 14+ MPa | 10-12 MPa | Extended durability |
| Latex Protein Content | Zero | High | No allergy complications |
These metrics provide defensible documentation for infection control committees and risk management teams. When facilities can demonstrate they selected gloves based on ASTM D6319 and EN 455 compliance rather than price alone, they strengthen their position against negligence claims.
Understanding what is a nitrile glove and its fundamental properties helps procurement teams make informed decisions. The material science behind nitrile rubber—a synthetic copolymer of butadiene and acrylonitrile—delivers the molecular structure necessary for superior barrier protection.
How Premium Nitrile Selection Reduces Healthcare-Associated Infection Liability Through Compliance
The Joint Commission, CDC, and WHO 2026 guidelines mandate task-specific glove selection to prevent HAIs and cross-contamination. These authoritative standards emphasize:
✅ Nonsterile nitrile for routine examinations
✅ Sterile nitrile for surgical procedures
✅ Heavy-duty 6-8 mil nitrile for extended chemical exposure
✅ Documented change protocols between patient contacts
Compliance with these guidelines requires facilities to maintain detailed traceability records showing:
- Batch numbers and manufacturing dates for all glove inventory
- Staff training completion on proper donning and doffing techniques
- Incident reports documenting glove failures or breaches
- Regular audits of glove selection appropriateness for specific tasks
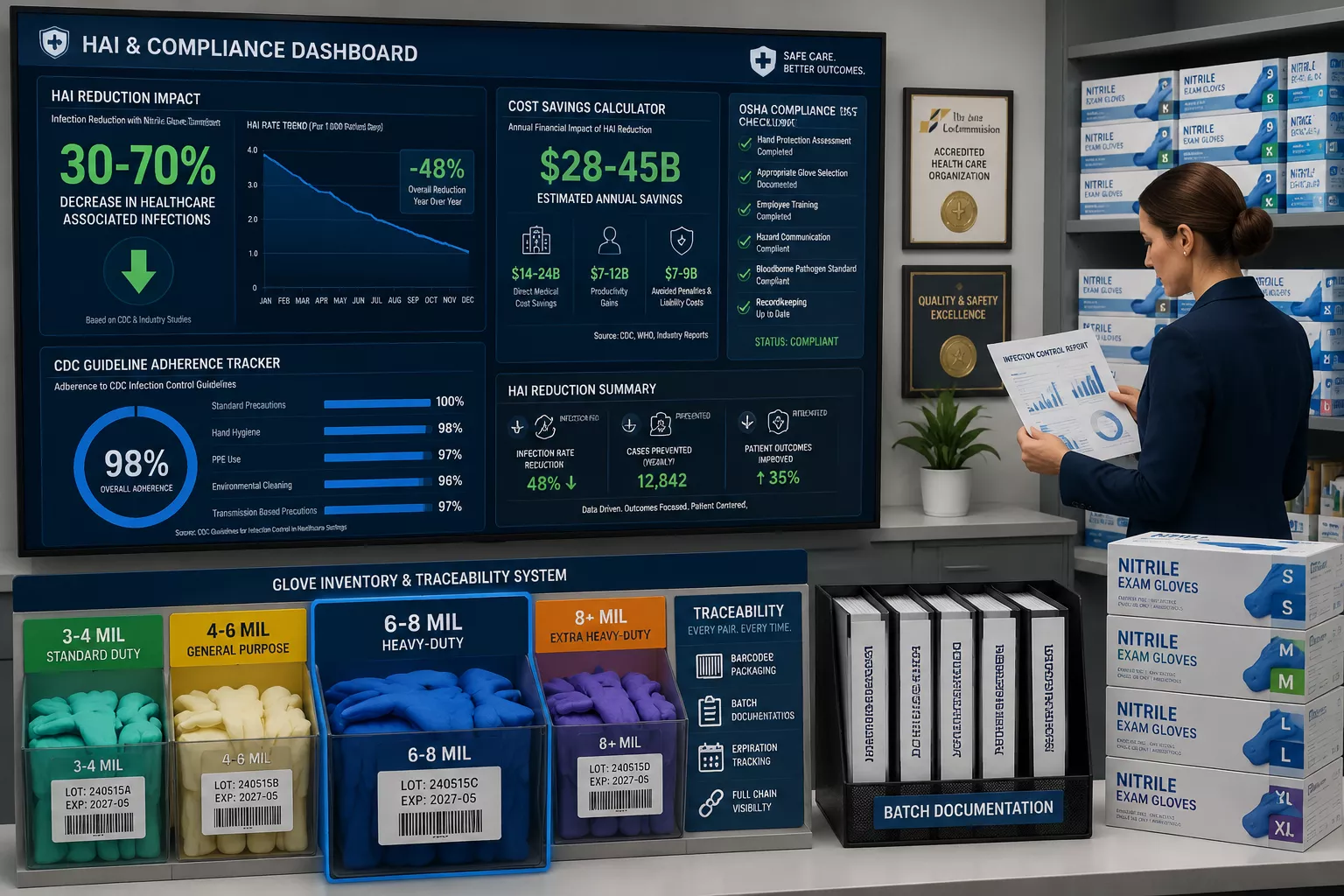
OSHA Compliance and Bloodborne Pathogen Standards
The Occupational Safety and Health Administration requires employers to provide appropriate personal protective equipment for bloodborne pathogen exposure risks. Premium nitrile gloves meeting FDA 510(k) clearance demonstrate compliance with these mandates.
Key OSHA compliance elements:
- Hazard assessment documentation showing why specific glove types were selected
- Employee training records on glove limitations and proper use
- Exposure control plans detailing glove change protocols
- Post-exposure evaluation procedures when glove failures occur
Facilities that document premium nitrile selection based on chemical permeation data and puncture resistance testing create a compliance trail that protects against citations and penalties. This documentation proves especially valuable during OSHA inspections or litigation discovery.
The importance of glove thickness selection cannot be overstated. Standard 4-5 mil nitrile suits routine examinations, while 6-8 mil versions provide necessary protection for chemotherapy drug handling, laboratory work, and extended procedures.
Chemical Resistance Data and Permeation Testing
Premium nitrile manufacturers provide detailed chemical resistance charts showing breakthrough times for specific substances. Berkeley Environmental Health & Safety recommends nitrile for incidental and extended contact with:
- Acids and bases (pH extremes)
- Petroleum products and oils 🛢️
- Alcohols and solvents
- Pharmaceutical compounds including controlled substances
This chemical resistance directly reduces exposure incidents that generate workers’ compensation claims and regulatory investigations. Facilities can reference manufacturer permeation data during incident reviews to demonstrate they provided appropriate protection.
Understanding what nitrile gloves protect against helps risk managers assess whether current glove selections adequately address facility-specific hazards. This knowledge proves critical when defending against claims of inadequate protective equipment.
Strategic Implementation: Maximizing HAI Reduction Through Premium Nitrile Programs
Even premium nitrile gloves fail to deliver their full protective potential without proper implementation. Healthcare facilities must address:
Training and Education Programs
Comprehensive staff education ensures the documented 30-70% HAI reduction potential becomes reality. Training should cover:
- Proper sizing selection to prevent tears from excessive stretching
- Donning techniques that avoid contamination during application
- Doffing procedures that prevent self-contamination during removal
- Change protocols specifying when gloves must be replaced
- Limitation awareness understanding that gloves degrade over time and with chemical exposure
Regular competency assessments verify that staff maintain proper technique. Video-based training modules with return demonstrations provide documented evidence of education completion.
Risk-Matched Selection Protocols
Different clinical environments require different glove specifications:
Emergency Department: 6 mil nitrile with textured grip for trauma situations
Operating Room: Sterile surgical nitrile with enhanced tactile sensitivity
Laboratory: Chemical-resistant nitrile rated for specific reagents
Patient Care Units: Standard 4-5 mil examination nitrile
Chemotherapy Preparation: 8 mil chemotherapy-tested nitrile with documented permeation data
Facilities that implement risk-matched selection protocols report fewer exposure incidents and better regulatory compliance. This systematic approach demonstrates thoughtful hazard assessment rather than one-size-fits-all purchasing.
For specialized applications, understanding alternatives to disposable nitrile gloves helps facilities evaluate whether reusable options might suit specific low-contamination tasks, though single-use remains the standard for patient care.
Quality Assurance and Vendor Management
Premium nitrile procurement requires vendor partnerships that provide:
- Consistent AQL ratings across all shipments (1.5 or lower preferred)
- Batch testing documentation showing compliance with ASTM standards
- Supply chain transparency ensuring authentic medical-grade products
- Technical support for chemical compatibility questions
- Recall procedures with rapid notification systems
Facilities should conduct periodic incoming quality inspections, testing random samples for pin-holes and tensile strength. This verification protects against counterfeit or substandard products that undermine infection control programs.
Awareness of potential nitrile glove allergies remains important, though true nitrile sensitivity affects less than 1% of users compared to 5-10% latex allergy rates. Facilities should maintain alternative options for the rare individual with confirmed nitrile sensitivity.
Cost-Benefit Analysis: Premium Investment vs. Liability Exposure
Healthcare CFOs increasingly recognize that premium nitrile gloves represent insurance against catastrophic infection outbreaks. A single HAI cluster can generate:
💰 $50,000-$150,000 in direct treatment costs per patient
⚖️ $500,000-$2M in litigation settlements
📉 10-30% reduction in patient volumes due to reputation damage
🚫 $10,000-$100,000 in regulatory penalties
When premium nitrile costs $0.08-$0.15 per glove compared to $0.05-$0.08 for standard options, the incremental investment of $0.03-$0.07 per glove becomes negligible against potential liability exposure. A 500-bed hospital using 5 million gloves annually spends an additional $150,000-$350,000 for premium protection—a fraction of a single serious HAI incident’s cost.
Return on Investment Calculations
Facilities can calculate ROI using this framework:
Annual Premium Cost = (Glove volume) × (Premium price – Standard price)
Avoided HAI Costs = (Baseline HAI rate – Post-implementation rate) × (Average HAI cost)
ROI = (Avoided costs – Premium investment) / Premium investment
Most facilities achieve positive ROI within the first year, with compounding benefits as infection rates decline and compliance improves. The 30-70% HAI reduction documented in clinical studies provides the foundation for these calculations.
Conclusion: Transforming Glove Selection Into Strategic Liability Management
Healthcare-Associated Infection Liability: How Premium Nitrile Glove Selection Reduces $28-45B Annual Costs represents more than a procurement decision—it’s a comprehensive risk management strategy. The documented performance advantages of premium nitrile gloves, including 3-4x greater puncture resistance, superior chemical barriers, and latex-free composition, directly address the infection control failures that generate billions in annual costs.
Immediate action steps for healthcare facilities:
- Audit current glove inventory against ASTM D6319 and EN 455 standards
- Implement risk-matched selection protocols with documented thickness and AQL requirements
- Establish comprehensive training programs covering proper donning, doffing, and change protocols
- Create traceability systems documenting batch numbers, training completion, and incident reports
- Partner with premium vendors who provide chemical permeation data and consistent quality
- Calculate facility-specific ROI using baseline HAI rates and avoided cost projections
The evidence overwhelmingly supports premium nitrile investment as both clinically effective and financially prudent. Facilities that document their selection criteria, maintain compliance records, and train staff properly position themselves to capture the full 30-70% HAI reduction potential while building defensible protection against liability claims.
As healthcare costs continue rising and regulatory scrutiny intensifies, the question isn’t whether facilities can afford premium nitrile gloves—it’s whether they can afford not to invest in proven infection control technology. The $28-45 billion annual HAI burden demands evidence-based solutions, and premium nitrile selection delivers measurable results that protect both patients and institutional financial health. 🏥