A single puncture in a chemical glove can expose skin to corrosive acids in under three seconds—yet nitrile gloves offer a critical advantage that latex and vinyl can’t match: visible tear detection. When nitrile rips, the damage is immediately obvious, giving lab workers and mechanics a split-second warning before disaster strikes. This transparency, combined with superior resistance to oils, greases, and many acids, has made understanding Nitrile Gloves in Chemical Handling: Protection Levels Against Oils, Acids, and Lab Hazards essential for anyone working with hazardous substances in 2026.
Whether you’re changing motor oil, handling laboratory solvents, or working with industrial chemicals, choosing the right nitrile glove thickness and understanding its limitations can mean the difference between safe handling and chemical burns. Let’s break down exactly what nitrile can—and can’t—protect you from.

Key Takeaways
- Nitrile gloves excel against oils, greases, and diluted acids but fail rapidly against concentrated nitric acid (>30%) and aromatic solvents like toluene
- Thickness matters tremendously: 3-5 mil gloves offer under 10 minutes protection against methylene chloride, while 8-15 mil versions provide extended exposure safety
- Temperature limits are strict: nitrile softens above 120°C (248°F) and can melt onto skin near flames or welding operations
- Visible tear detection gives nitrile a safety edge over latex—rips and punctures are immediately obvious
- Always consult chemical compatibility charts before use; not all nitrile formulations protect against every chemical
Understanding Nitrile Gloves in Chemical Handling: What Makes Them Different
Nitrile gloves aren’t just “better latex”—they’re a completely different synthetic rubber (acrylonitrile-butadiene) engineered specifically for chemical resistance. While latex gloves have their place in healthcare settings, nitrile dominates chemical handling for three key reasons:
🛢️ Superior Oil and Grease Resistance
Unlike latex, which breaks down rapidly when exposed to petroleum products, nitrile maintains its integrity against motor oils, hydraulic fluids, and industrial greases. This makes it the go-to choice for automotive mechanics handling greasy components and manufacturing workers dealing with lubricants.
🧪 Broader Chemical Compatibility
Nitrile resists a wider spectrum of chemicals including many solvents, diluted acids, and bases. The synthetic rubber structure doesn’t react with most organic compounds the way natural latex does.
💪 Puncture and Tear Resistance
Nitrile is significantly tougher than latex or vinyl. When comparing nitrile to vinyl gloves in durability tests, nitrile consistently shows 3-5 times greater puncture resistance—critical when handling sharp laboratory equipment or rough mechanical parts.
Hypoallergenic Properties
For workers with latex allergies, nitrile provides comparable protection without the allergenic proteins found in natural rubber. This has made it the standard in laboratories and industrial settings where latex sensitivity is a concern.
Protection Levels: Nitrile Gloves Against Oils, Acids, and Common Lab Chemicals
Not all chemical exposures are created equal, and neither are all nitrile gloves. Understanding the protection levels of Nitrile Gloves in Chemical Handling: Protection Levels Against Oils, Acids, and Lab Hazards requires looking at both the chemical type and glove specifications.
Oils and Petroleum Products: Where Nitrile Shines ✅
Nitrile gloves provide excellent resistance to:
- Motor oils and transmission fluids
- Hydraulic oils
- Gasoline and diesel fuel (short-term exposure)
- Mineral oils
- Vegetable oils and animal fats
- Industrial greases and lubricants
For petroleum-based oil handling, even standard 4-6 mil disposable nitrile gloves offer adequate splash protection. For extended contact, 8 mil or thicker reusable nitrile gloves are recommended. The oil resistance comes from nitrile’s non-polar molecular structure, which doesn’t interact with hydrocarbon chains.
Acids: Strong Performance with Critical Exceptions ⚠️
Nitrile handles well:
- Diluted sulfuric acid (<30%)
- Diluted hydrochloric acid
- Acetic acid
- Phosphoric acid
- Citric acid and other weak organic acids
Nitrile fails against:
- Concentrated nitric acid (>30%): Causes rapid degradation, often within minutes
- Fuming sulfuric acid (oleum): Destroys nitrile almost immediately
- Highly oxidizing acids: Break down the polymer chains
For concentrated acids, switch to neoprene or thick laminate gloves specifically rated for strong oxidizers. Always check the Safety Data Sheet (SDS) for the specific acid concentration you’re handling.
Solvents: The Tricky Category 🔬
Nitrile’s solvent resistance varies dramatically:
| Solvent Type | Nitrile Performance | Breakthrough Time (5 mil) |
|---|---|---|
| Alcohols (ethanol, isopropanol) | Excellent | >8 hours |
| Ketones (acetone, MEK) | Fair to Poor | 10-30 minutes |
| Chlorinated (methylene chloride) | Poor | <10 minutes |
| Aromatics (toluene, xylene, benzene) | Poor | <20 minutes |
| Aliphatics (hexane, heptane) | Good | 2-4 hours |
For handling hazardous solvents like acetone, thin disposable nitrile provides only brief splash protection. Thicker 8-15 mil nitrile performs better but still isn’t recommended for prolonged aromatic solvent exposure. For these chemicals, consider butyl rubber or specialized laminate gloves.
Bases and Caustics: Generally Good Protection ✅
Nitrile resists most bases effectively:
- Sodium hydroxide (lye) solutions
- Potassium hydroxide
- Ammonia solutions
- Calcium hydroxide
Standard 5-6 mil nitrile gloves provide adequate protection for typical laboratory concentrations of these chemicals.
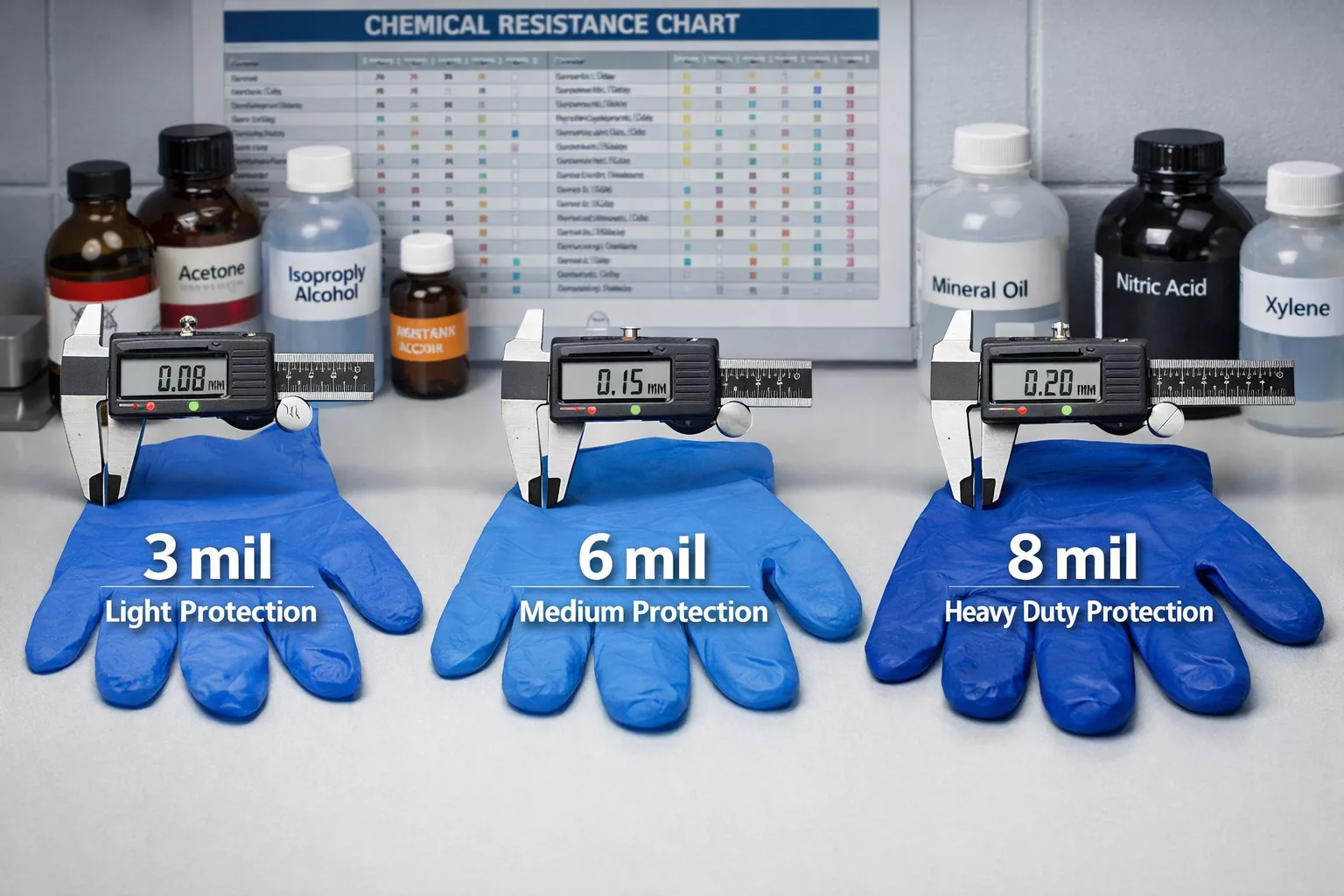
Thickness Matters: Choosing the Right Mil Rating for Chemical Handling
When evaluating Nitrile Gloves in Chemical Handling: Protection Levels Against Oils, Acids, and Lab Hazards, thickness is perhaps the most critical specification. The “mil” measurement (1 mil = 0.001 inch) directly correlates with breakthrough time and durability.

Thin Disposable (3-5 mil): Laboratory Examination Gloves
Best for:
- Brief chemical contact and splash protection
- General laboratory work with low-hazard chemicals
- Tasks requiring high dexterity and tactile sensitivity
- Frequent glove changes
Limitations:
- Breakthrough time under 10 minutes for aggressive solvents
- Limited puncture resistance
- Not suitable for extended chemical exposure
These are the standard gloves found in most lab dispensers. They’re designed for incidental contact only—change them immediately if contaminated. For more guidance, see our article on what thickness is best for different applications.
Medium Weight (6-8 mil): Industrial Chemical Handling
Best for:
- Extended exposure to oils and greases
- Industrial cleaning with chemical solutions
- Automotive work with fuels and lubricants
- Manufacturing processes involving chemicals
Advantages:
- 2-4x longer breakthrough times than thin gloves
- Better puncture and abrasion resistance
- Suitable for tasks lasting 30 minutes to several hours
This thickness range offers the best balance between protection and dexterity for most industrial chemical handling tasks. Understanding physical factors that affect breakthrough time helps optimize glove selection.
Heavy Duty (8-15 mil): Reusable Chemical Gloves
Best for:
- Prolonged immersion in chemicals
- Heavy industrial applications
- High-concentration chemical handling
- Situations requiring maximum protection
Trade-offs:
- Reduced tactile sensitivity
- Less dexterity for fine motor tasks
- Higher cost (but reusable if properly maintained)
For serious chemical work, these thicker gloves are essential. They’re often sold as reusable gloves with extended cuffs for forearm protection.
Temperature Limitations: When Nitrile Isn’t Enough 🔥
One critical limitation often overlooked: nitrile gloves soften and lose integrity above 120°C (248°F). This creates serious hazards in specific situations:
Never use nitrile gloves for:
- Welding or torch work
- Handling objects near open flames
- High-temperature chemical processes
- Autoclave operations (though some specialized nitrile can be autoclaved once)
The danger isn’t just loss of protection—melted nitrile can adhere to skin, causing severe burns. For high-heat applications, use leather, Kevlar, or specialized heat-resistant gloves instead.
On the cold end, nitrile remains flexible down to approximately -40°C, making it suitable for cold storage and refrigerated environments.
Standards and Certifications: Decoding the Labels 📋
Professional chemical handling gloves should meet specific standards:
EN ISO 374-1: Chemical permeation resistance
- Type A: Passes breakthrough test for at least 6 chemicals (>30 min each)
- Type B: Passes for at least 3 chemicals
- Type C: Passes for at least 1 chemical (>10 min)
EN ISO 374-5: Protection against microorganisms and chemicals
ASTM D6978: Standard practice for assessing chemical resistance
Always check the manufacturer’s chemical compatibility chart before use. These charts list specific chemicals and the tested breakthrough times for that particular glove formulation. Not all nitrile is created equal—different manufacturing processes and additives affect chemical resistance.
Best Practices for Using Nitrile Gloves in Chemical Handling 🧤
Even the best gloves fail if used incorrectly. Follow these protocols to maximize protection:
Before Use
- Inspect every glove for tears, punctures, or discoloration before donning
- Check the expiration date—nitrile degrades over time, especially if exposed to ozone or UV light
- Verify chemical compatibility using the manufacturer’s chart
- Select appropriate thickness for the exposure duration and chemical type
During Use
- Never reuse disposable nitrile gloves after chemical contact
- Change gloves immediately if you notice any discoloration, swelling, or stiffness
- Avoid touching your face, phone, or other surfaces while wearing contaminated gloves
- Use double-gloving for high-hazard chemicals (outer glove for protection, inner for backup)
- Extend cuffs over lab coat sleeves to prevent chemicals from running down your arms
After Use
- Remove gloves properly using the inside-out technique to avoid skin contact with the contaminated exterior
- Wash hands thoroughly even after glove use—no glove provides 100% protection
- Dispose in appropriate chemical waste containers—contaminated gloves are hazardous waste
- Never leave used gloves on lab benches where others might contact them
For workers concerned about environmental impact, check out our guide on whether nitrile gloves can be recycled.
Common Mistakes That Compromise Protection ❌
Using the wrong glove for the chemical: Just because it’s nitrile doesn’t mean it protects against everything. Always verify compatibility.
Wearing gloves too long: Breakthrough can occur invisibly—follow recommended maximum wear times for your specific chemicals.
Ignoring visible damage: That small tear might seem minor, but it creates a direct pathway for chemicals to reach your skin.
Assuming all nitrile is the same: Manufacturing quality, thickness, and formulation vary significantly between brands and product lines.
Forgetting about permeation: Chemicals can pass through intact gloves at the molecular level without visible damage—this is why breakthrough time matters.
Alternatives When Nitrile Isn’t Appropriate 🔄
For chemicals that degrade nitrile, consider these alternatives:
- Butyl rubber: Excellent for ketones, esters, and highly polar solvents
- Neoprene: Superior for oxidizing acids and some chlorinated solvents
- Viton (fluoroelastomer): Outstanding aromatic and chlorinated solvent resistance
- Laminate gloves: Multi-layer construction for broad chemical protection
- Silver Shield: Thin laminate offering protection against 280+ chemicals
Each material has its own strengths and weaknesses. For specialized applications like resin printing, specific glove types may be recommended.
Conclusion: Making Smart Choices for Chemical Safety
Understanding Nitrile Gloves in Chemical Handling: Protection Levels Against Oils, Acids, and Lab Hazards isn’t just about memorizing compatibility charts—it’s about developing a safety mindset that prioritizes proper glove selection, usage, and disposal. Nitrile gloves offer exceptional protection against oils, greases, many acids, and numerous laboratory chemicals, but they’re not a universal solution.
Your action plan for 2026:
- Audit your chemical inventory and create a glove selection matrix matching each chemical to the appropriate glove type and thickness
- Train all personnel on proper donning, doffing, and inspection procedures
- Post chemical compatibility charts at every chemical storage area and workstation
- Establish maximum wear times for different chemical exposures
- Implement a double-gloving protocol for high-hazard chemicals
- Review and update your glove selection as new chemicals are introduced
Remember: the visible tear detection that makes nitrile so valuable only works if you’re actually looking at your gloves during use. Make visual inspection a habit, change gloves at the first sign of damage or chemical contact, and never assume protection without verification.
The right nitrile glove, properly selected and used, provides outstanding chemical protection. The wrong glove—or the right glove used incorrectly—provides a dangerous false sense of security. Know your chemicals, know your gloves, and always prioritize safety over convenience.