A single particle measuring just 0.5 micrometers can destroy an entire semiconductor wafer worth thousands of dollars. In 2026, the nitrile cleanroom gloves market has reached $375.9 million, driven by pharmaceutical, biotech, and semiconductor industries where contamination control isn’t just important—it’s everything. Nitrile Gloves in Cleanroom Environments: Standards, Selection, and Contamination Prevention Guide has become essential reading for quality managers, cleanroom operators, and procurement specialists navigating increasingly strict contamination protocols.
The shift from latex to nitrile in controlled environments reflects more than allergy concerns. With nitrile gloves now commanding 47.8% of the cleanroom gloves market, understanding ASTM and ISO standards, particle shedding rates, and application-specific requirements separates compliant operations from costly contamination events.

Key Takeaways
- ISO 14644 and ASTM D6319 standards define particle limits and testing protocols for cleanroom nitrile gloves across different classification levels
- Nitrile gloves dominate cleanrooms with superior chemical resistance, puncture protection, and latex-free composition ideal for pharma, biotech, and semiconductor applications
- Particle shedding rates vary dramatically by glove thickness, manufacturing process, and cleanroom class—requiring careful matching to environment specifications
- Double-gloving protocols and proper donning techniques reduce contamination risk by up to 85% in critical manufacturing zones
- 2026 regulatory updates emphasize continuous monitoring, digital audit trails, and risk-based ESD protection for cleanroom glove programs
Understanding Cleanroom Standards for Nitrile Gloves
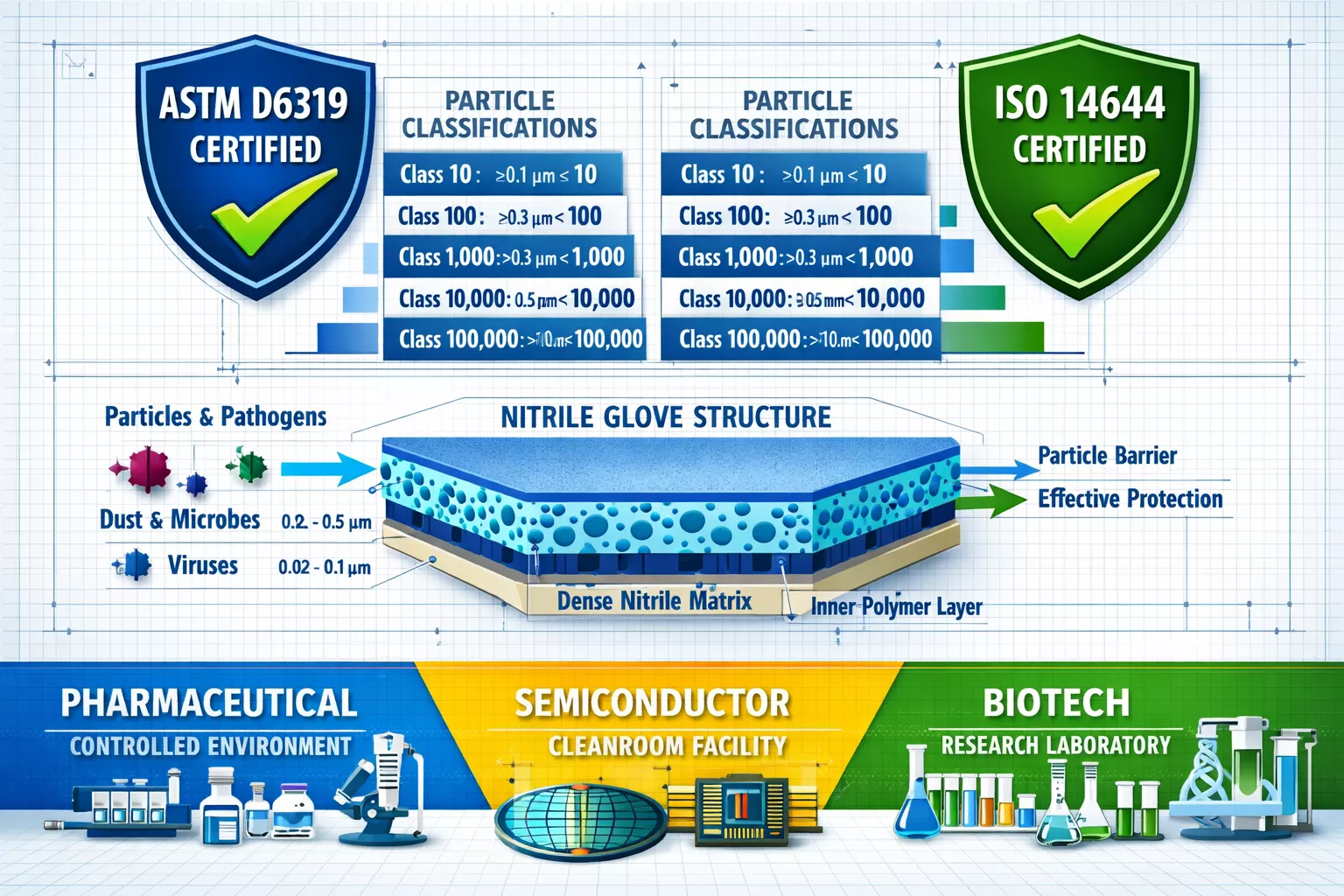
ISO 14644 Classifications and Particle Limits
ISO 14644 establishes the global framework for cleanroom classification based on airborne particle concentration. For nitrile gloves, this standard directly impacts selection criteria across different cleanroom classes:
Cleanroom Classification Table
| ISO Class | Particles ≥0.5μm per m³ | Typical Applications | Nitrile Glove Requirements |
|---|---|---|---|
| ISO 3 | 35 | Semiconductor lithography | Ultra-low particle shedding, Class 10 rated |
| ISO 5 | 3,520 | Pharmaceutical aseptic filling | Class 100 rated, sterile packaging |
| ISO 7 | 352,000 | Medical device assembly | Class 10,000 rated, powder-free |
| ISO 8 | 3,520,000 | General pharmaceutical manufacturing | Class 100,000 rated, chemical resistant |
The 2026 regulatory landscape maintains ISO 14644 core requirements while emphasizing continuous particle monitoring and documented standard operating procedures. Cleanroom managers must now demonstrate real-time compliance rather than periodic testing alone.
ASTM D6319: The Nitrile Glove Testing Standard
ASTM D6319 specifically addresses cleanroom gloves, establishing test methods for:
- Particle generation rates through tumble testing
- Extractable contamination levels in solvents
- Physical integrity including pinhole detection
- Chemical resistance to common cleanroom agents
Gloves meeting ASTM D6319 undergo rigorous tumble testing where particle counts are measured after mechanical agitation. Quality nitrile cleanroom gloves typically shed fewer than 300 particles ≥0.5μm during standardized testing—critical for ISO Class 5 and cleaner environments.
FDA and ESD Compliance Requirements
Beyond particle control, 2026 regulations integrate multiple compliance layers:
FDA Alignment: The FDA now mandates alignment with ISO 14644 and EU Annex 1 for pharmaceutical cleanrooms, requiring:
- Unidirectional airflow in Grade A zones
- Semi-annual media fills for aseptic processes
- Documented glove qualification protocols
ESD Protection: ANSI/ESD S20.20 updates stress risk-based approaches for electronics manufacturing. ESD-safe nitrile gloves must demonstrate surface resistivity between 10⁴ and 10¹¹ ohms, preventing electrostatic discharge that damages sensitive components.
Understanding what makes nitrile gloves suitable for specialized applications helps cleanroom managers balance multiple compliance requirements simultaneously.
Selecting the Right Nitrile Gloves for Cleanroom Applications
Thickness Considerations by Industry
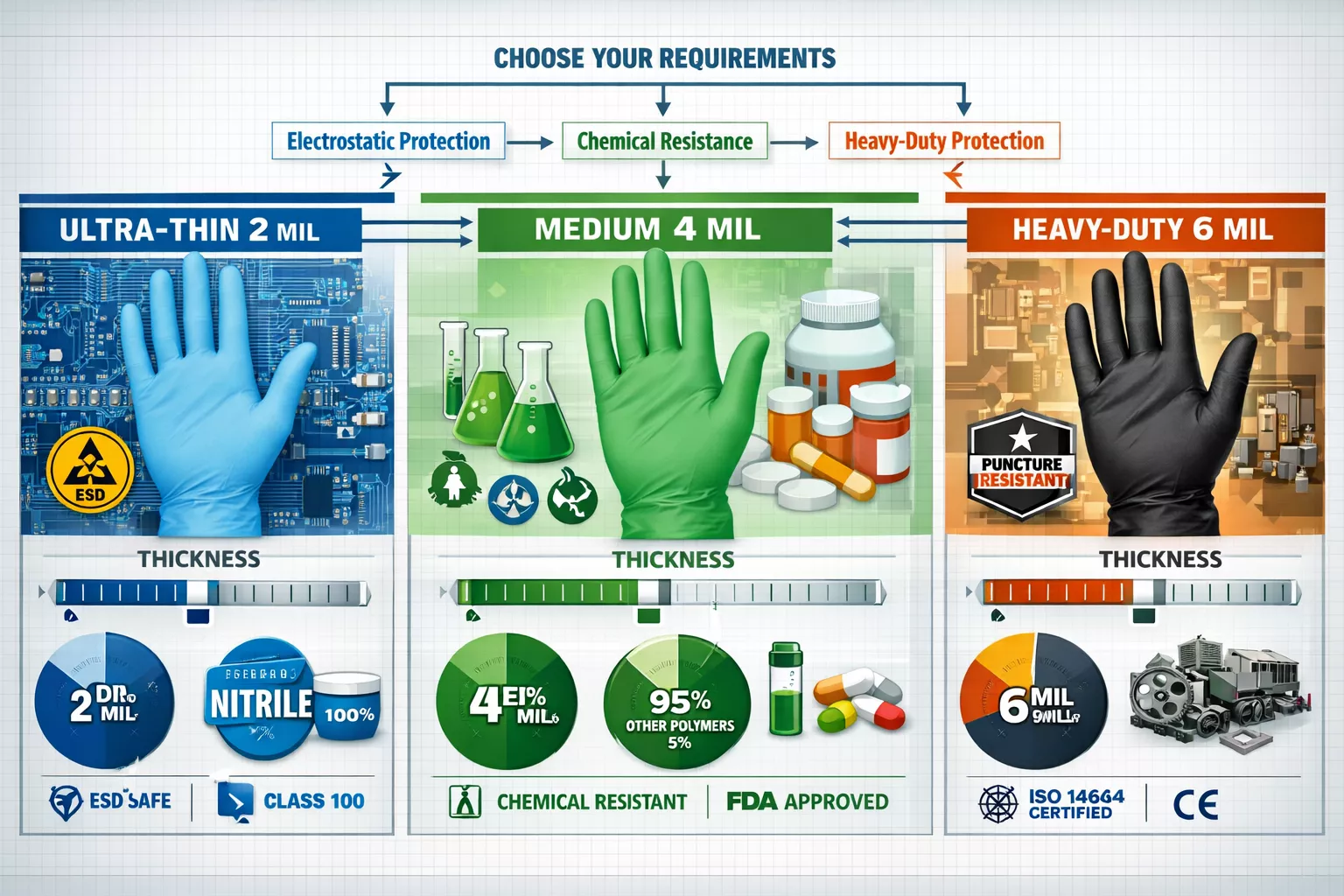
Nitrile glove thickness dramatically affects both protection and particle generation:
Semiconductor Manufacturing (ISO 3-5):
- 2-3 mil thickness provides maximum dexterity for microelectronics handling
- Ultra-smooth surface finish minimizes particle shedding
- ESD-safe formulations prevent component damage
- Recent innovations include halogen-free variants launched by Top Glove in July 2025 for aerospace applications
Pharmaceutical Production (ISO 5-7):
- 4-5 mil thickness balances chemical resistance with tactile sensitivity
- Sterile packaging essential for aseptic processing zones
- Chemotherapy-tested variants now available for oncology drug manufacturing
- Understanding glove thickness impact on durability becomes crucial for extended wear periods
Biotech Research (ISO 6-8):
- 5-6 mil thickness offers enhanced puncture resistance for sharps handling
- Extended cuff lengths (12+ inches) protect forearms during biological work
- Chemical resistance to common lab solvents and disinfectants
The optimal thickness for nitrile gloves depends on balancing protection requirements against particle generation risks—thicker gloves generally shed more particles during movement.
Material Composition and Chemical Resistance
Modern cleanroom nitrile gloves feature specialized formulations:
Polymer Blends: Advanced nitrile butadiene rubber (NBR) formulations incorporate:
- Accelerator-free chemistry reducing extractable contaminants
- Low-modulus compounds improving comfort during extended wear
- Biodegradable additives addressing environmental concerns (Shield Scientific’s October 2025 launch)
Chemical Compatibility: Nitrile excels against:
- ✅ Alcohols (isopropanol, ethanol cleaning agents)
- ✅ Weak acids and bases common in pharmaceutical processing
- ✅ Petroleum-based solvents in semiconductor manufacturing
- ✅ Disinfectants including quaternary ammonium compounds
However, nitrile shows limited resistance to:
- ❌ Ketones (acetone, MEK)
- ❌ Strong oxidizing acids
- ❌ Aromatic hydrocarbons at high concentrations
The composition of nitrile gloves directly influences both contamination control and worker safety in chemical-intensive cleanroom processes.
Sterile vs. Non-Sterile Cleanroom Gloves
Sterile Nitrile Gloves:
- Gamma or e-beam irradiation sterilization
- Individual or double-bagged packaging
- Required for ISO Class 5 aseptic zones
- Higher cost ($0.50-$2.00 per pair)
- Expiration dating and lot traceability
Non-Sterile Cleanroom Gloves:
- Manufactured in controlled environments
- Bulk packaging in cleanroom-compatible bags
- Suitable for ISO Class 6-8 environments
- Cost-effective ($0.15-$0.50 per pair)
- Focus on particle control rather than microbial sterility
Understanding the difference between sterile and non-sterile gloves prevents over-specification that inflates costs without improving contamination control.
Recent Product Innovations (2025-2026)
The cleanroom glove market has seen significant advancement:
Ansell’s Kimtech Polaris (June 2025): Launched in Japan specifically for non-sterile pharmaceutical and biotech cleanrooms, offering enhanced chemical resistance and ergonomic design for Asian market hand sizes.
Top Glove Halogen-Free Series (July 2025): Addresses semiconductor industry concerns about halogen contamination in advanced chip manufacturing, particularly critical for sub-7nm processes.
Shield Scientific High-Performance Line (October 2025): Two new nitrile formulations reducing heat stress during extended wear while maintaining particle barrier compliance—addressing worker comfort complaints that led to protocol violations.
These innovations reflect industry pressure for gloves that simultaneously improve contamination control, worker comfort, and environmental sustainability.
Contamination Prevention Protocols and Best Practices
Proper Donning and Doffing Techniques
The Five-Step Cleanroom Gloving Protocol:
- Hand Hygiene: Wash with cleanroom-approved soap, dry completely with lint-free towels
- First Glove Application: Touch only the cuff interior, pull over hand without touching exterior surfaces
- Second Glove Application (if double-gloving): Use gloved hand to handle second glove, overlap cuffs by 2-3 inches
- Inspection: Check for visible defects, tears, or contamination under cleanroom lighting
- Sanitization: Spray with 70% isopropanol, allow to air dry before entering critical zones
Common Donning Errors that compromise contamination control:
- 🚫 Touching glove exterior with bare hands
- 🚫 Inadequate cuff overlap in double-gloving
- 🚫 Failing to inspect for manufacturing defects
- 🚫 Entering cleanroom before gloves fully dry post-sanitization
Double-Gloving Strategies
Double-gloving reduces contamination risk by up to 85% in critical manufacturing zones:
Pharmaceutical Aseptic Processing:
- Inner glove: 4-mil sterile nitrile, changed every 2 hours
- Outer glove: 5-mil sterile nitrile, changed after each critical intervention
- Color differentiation (blue inner, purple outer) enables visual breach detection
Semiconductor Wafer Handling:
- Inner glove: 2-mil ESD-safe nitrile
- Outer glove: 3-mil ultra-low particle shedding nitrile
- Both gloves changed between lot processing to prevent cross-contamination
Biotech Cell Culture:
- Inner glove: 4-mil powder-free nitrile
- Outer glove: 5-mil extended cuff nitrile
- Outer glove sanitized between hood entries, both changed every 4 hours
The healthcare demand for nitrile gloves has driven innovations in double-gloving comfort that translate directly to cleanroom applications.

Particle Shedding Minimization
Manufacturing Process Impact:
- Chlorinated gloves: Surface treatment reduces particle shedding by 40-60%
- Polymer washing: Removes residual manufacturing chemicals that attract particles
- Smooth finish: Textured surfaces shed 2-3x more particles than smooth alternatives
Operational Practices:
- Limit hand movements in ISO Class 5 and cleaner zones
- Use slow, deliberate motions rather than rapid gestures
- Replace gloves every 2-4 hours even without visible contamination
- Store gloves in original cleanroom-compatible packaging until use
Environmental Controls:
- Maintain 45-55% relative humidity (too dry increases static, too humid promotes microbial growth)
- Ensure proper HEPA filtration with 90+ air changes per hour
- Implement unidirectional airflow in critical zones
- Monitor particle counts continuously rather than periodically
Supply Chain Challenges in 2026
Despite market growth, persistent nitrile glove shortages continue affecting cleanroom operations. U.S. federal orders alone exceed 55.5 million boxes, outstripping nitrile butadiene rubber (NBR) supply capacity.
Mitigation Strategies:
- 📦 Maintain 90-day inventory buffers for critical glove specifications
- 🤝 Establish relationships with multiple qualified suppliers
- 📊 Implement just-in-time-plus inventory management
- 🔄 Qualify alternative glove models before shortages occur
- 📝 Document equivalency testing for regulatory compliance
The future of disposable gloves includes increased domestic manufacturing capacity and biodegradable formulations addressing both supply security and environmental concerns.
Quality Management System Integration
ISO 9001:2026 revisions emphasize digital transformation in quality management:
Digital Audit Trails: Electronic batch records must document:
- Glove lot numbers used in each manufacturing run
- Change-out times and personnel identification
- Particle count data correlated with glove usage
- Non-conformance events and corrective actions
Risk Management: Failure Mode and Effects Analysis (FMEA) for glove selection should evaluate:
- Particle generation risk vs. cleanroom class
- Chemical breakthrough risk vs. process chemicals
- Allergic reaction risk vs. workforce sensitivities (though nitrile is generally hypoallergenic)
- Supply chain disruption risk vs. inventory costs
Supplier Oversight: Enhanced requirements for vendor qualification including:
- Annual audits of manufacturing facilities
- Certificate of Analysis (CoA) for each lot received
- Particle shedding test data per ASTM D6319
- Chemical resistance validation for specific applications
Conclusion
Nitrile Gloves in Cleanroom Environments: Standards, Selection, and Contamination Prevention Guide represents a critical knowledge area as contamination control requirements intensify across pharmaceutical, biotech, and semiconductor industries. The 47.8% market share nitrile commands in cleanroom applications reflects its superior balance of chemical resistance, particle control, and allergy safety compared to latex alternatives.
Success in cleanroom glove management requires understanding the interplay between ISO 14644 classifications, ASTM D6319 testing protocols, and application-specific requirements. Semiconductor facilities demand ultra-thin, ESD-safe gloves with minimal particle shedding, while pharmaceutical aseptic processing requires sterile gloves with proven chemical resistance to cleaning agents and drug compounds.
Actionable Next Steps
✅ Audit Current Glove Specifications: Compare existing gloves against ASTM D6319 particle generation data and ISO cleanroom class requirements
✅ Implement Double-Gloving Protocols: Establish color-coded systems for critical zones with documented change-out frequencies
✅ Qualify Multiple Suppliers: Address ongoing supply chain challenges by validating alternative glove sources before shortages occur
✅ Upgrade Quality Systems: Integrate digital audit trails and risk-based supplier oversight per ISO 9001:2026 requirements
✅ Train Personnel Regularly: Conduct quarterly refresher training on proper donning, doffing, and contamination prevention techniques
✅ Monitor Regulatory Updates: Track FDA, ISO, and ANSI/ESD standard revisions affecting cleanroom glove requirements
The $375.9 million nitrile cleanroom gloves market will continue growing at 4.7% CAGR through 2034, driven by stricter contamination controls and expanding applications in emerging technologies. Organizations that master the technical nuances of glove selection, implement robust contamination prevention protocols, and maintain agile supply chains will gain competitive advantages in quality, compliance, and operational efficiency.
Whether managing an ISO Class 3 semiconductor fab or an ISO Class 7 pharmaceutical packaging line, the right nitrile glove selection—backed by proper protocols and continuous improvement—transforms contamination control from a compliance burden into a strategic quality advantage. 🧤✨