A single particle measuring just 0.5 microns can compromise an entire batch of sterile pharmaceuticals worth millions of dollars. In 2026, the global nitrile cleanroom gloves market has reached USD 375.9 million, driven by one critical factor: the pharmaceutical industry’s zero-tolerance approach to contamination. Nitrile Gloves in Pharmaceuticals: Compliance with GMP Standards and Particle Control isn’t just about hand protection—it’s about safeguarding drug efficacy, patient safety, and regulatory compliance in an industry where microscopic failures have massive consequences.
The pharmaceutical sector demands more than basic barrier protection. Every glove worn in drug manufacturing must meet stringent Good Manufacturing Practice (GMP) standards while generating minimal particles that could contaminate sterile products. With powder-free nitrile gloves now holding 73.9% of the market share and regulatory bodies tightening cleanroom requirements, understanding how to select and use low-particulate nitrile gloves has become essential for pharmaceutical operations.
Key Takeaways
- Powder-free nitrile gloves are mandatory in pharmaceutical cleanrooms to prevent particle contamination and meet GMP compliance standards
- The nitrile cleanroom gloves market is projected to reach USD 490 million by 2034, fueled by pharmaceutical expansion and contamination control regulations
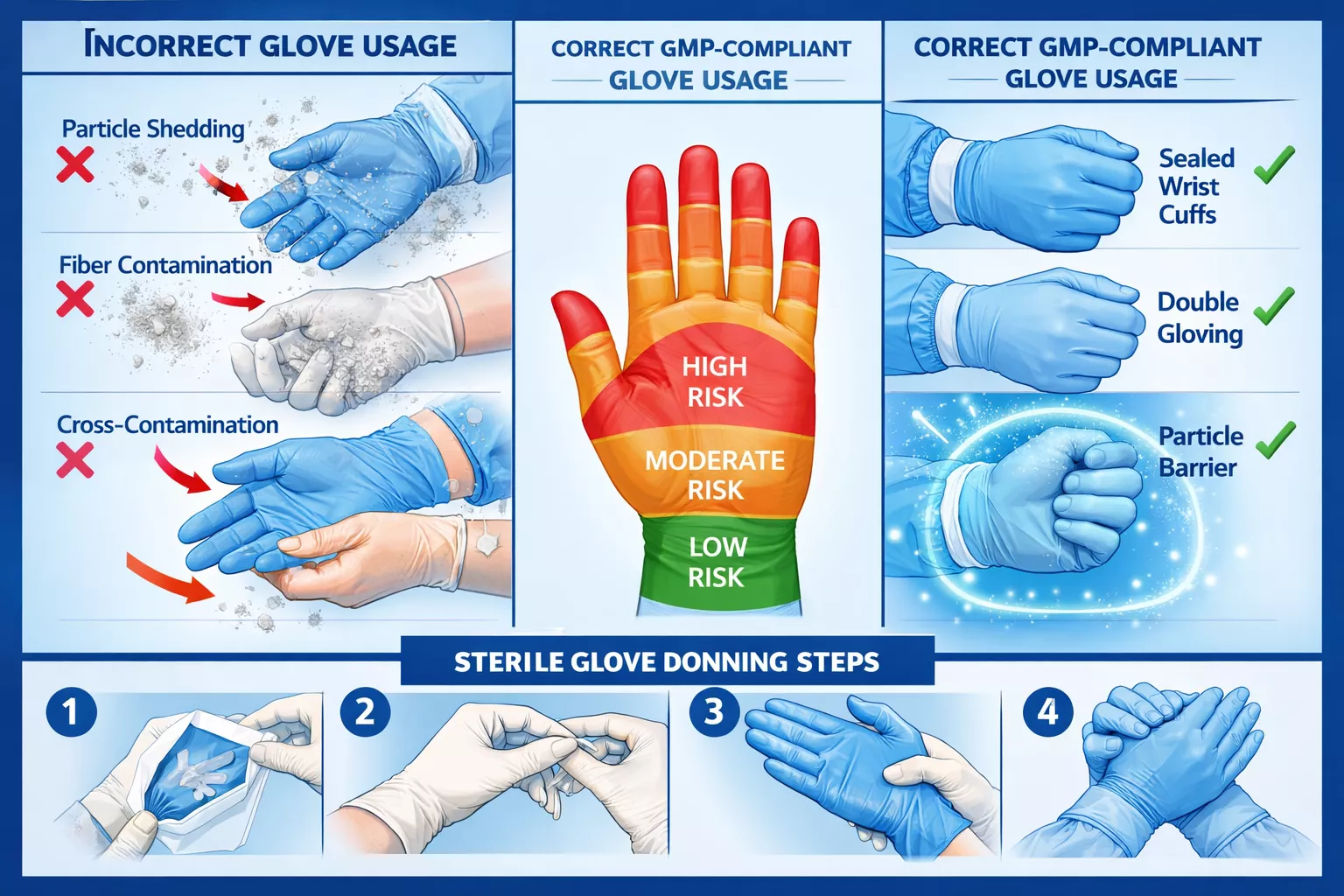
- ISO Class 1-5 cleanroom-rated nitrile gloves generate fewer than 300 particles per glove, critical for sterile drug manufacturing
- Nitrile offers superior chemical resistance and is 100% latex-free, eliminating allergic reactions while maintaining tactile sensitivity for precision work
- Proper glove selection, donning procedures, and change protocols are essential to avoid cross-contamination in pharmaceutical environments

Understanding GMP Standards for Nitrile Gloves in Pharmaceuticals
Good Manufacturing Practice (GMP) standards establish the baseline for pharmaceutical production quality. When it comes to Nitrile Gloves in Pharmaceuticals: Compliance with GMP Standards and Particle Control, these regulations dictate everything from material composition to particle generation limits.
What Makes a Glove GMP-Compliant?
GMP-compliant nitrile gloves must meet several critical criteria:
- Powder-free formulation 🚫 – Cornstarch powder used in older gloves generates particles and can contaminate products
- Low particle generation – Cleanroom-grade gloves produce fewer than 300 particles (≥0.5 microns) per glove
- Chemical resistance – Must withstand pharmaceutical solvents, disinfectants, and active ingredients without degradation
- Sterility assurance – For aseptic processing, gloves must be gamma-irradiated or undergo validated sterilization
- Traceability – Batch numbers and quality certificates ensure accountability
The shift away from latex gloves has been dramatic. Nitrile gloves are safe for latex allergy sufferers, making them the default choice in pharmaceutical facilities where worker safety and product integrity both matter.
Regulatory Frameworks Driving Compliance
Multiple regulatory bodies enforce strict standards:
FDA (United States): Requires pharmaceutical manufacturers to use appropriate protective equipment that doesn’t compromise product quality. Current Good Manufacturing Practice (cGMP) regulations mandate contamination control measures.
EMA/EU GMP Annex 1: The European Medicines Agency’s Annex 1 guidelines for sterile medicinal products specifically address cleanroom garments and gloves, requiring validation of particle generation and sterility.
ISO 14644 Standards: Define cleanroom classifications from ISO Class 1 (ultra-clean) to ISO Class 9, with corresponding particle count limits that gloves must not exceed.
Companies like Ansell have developed specialized nitrile gloves specifically for Annex 1 compliance, focusing on minimizing particle shedding during critical aseptic operations.
Particle Control: Why Low-Particulate Nitrile Gloves Matter
Particle contamination represents one of the most significant risks in pharmaceutical manufacturing. Even microscopic particles can:
- Introduce microbial contamination into sterile products 🦠
- Cause adverse reactions in injectable medications
- Reduce drug efficacy through chemical interactions
- Trigger costly batch rejections and regulatory actions
The Science Behind Particle Generation
Traditional gloves shed particles through several mechanisms:
- Friction – Movement between glove surfaces generates static electricity and releases material fragments
- Degradation – Chemical exposure breaks down glove polymers, creating micro-particles
- Manufacturing residues – Poor quality control leaves loose fibers and material debris
- Powder additives – Cornstarch particles become airborne and settle on surfaces
Low-particulate nitrile gloves address these issues through:
- Advanced polymer formulation with tighter molecular bonds
- Cleanroom manufacturing environments (often ISO Class 7 or better)
- Double-bagging in sealed, particle-free packaging
- Post-production washing to remove surface contaminants
Cleanroom Classification Requirements
Different pharmaceutical operations require different cleanroom classes:
| Cleanroom Class | Particles/m³ (≥0.5 μm) | Pharmaceutical Application | Glove Requirements |
|---|---|---|---|
| ISO Class 3 | 35 | Aseptic filling, critical zones | Ultra-low particle, sterile |
| ISO Class 5 | 3,520 | Sterile product preparation | Low particle, sterile |
| ISO Class 7 | 352,000 | Non-sterile compounding | Low particle, powder-free |
| ISO Class 8 | 3,520,000 | Packaging, warehousing | Standard powder-free |
The tighter the cleanroom classification, the more critical glove selection becomes. Understanding how long nitrile gloves last helps facilities plan change protocols that maintain contamination control without excessive waste.

Selecting the Right Nitrile Gloves for Pharmaceutical Applications
Not all nitrile gloves are created equal. Nitrile Gloves in Pharmaceuticals: Compliance with GMP Standards and Particle Control requires careful selection based on specific operational needs.
Key Selection Criteria
Thickness and Durability ⚡
Pharmaceutical work demands a balance between protection and tactile sensitivity:
- 4-5 mil thickness – Standard for general pharmaceutical handling, offering good dexterity
- 6-8 mil thickness – Enhanced protection for chemical handling and extended wear
- Double-gloving – Common in sterile compounding, with thinner inner glove (4 mil) and thicker outer glove (6 mil)
Chemical Resistance Profile
Nitrile excels against many pharmaceutical chemicals, but specific formulations vary. Consider resistance to:
- Alcohols (isopropanol, ethanol) used in disinfection
- Organic solvents (acetone, methanol, toluene)
- Acids and bases used in synthesis
- Oils and petroleum-based compounds
Nitrile gloves protect against various types of oils, making them versatile for pharmaceutical manufacturing environments.
Texture and Grip
Pharmaceutical tasks require precision handling of small vials, syringes, and instruments:
- Textured fingertips – Enhanced grip for wet or slippery surfaces
- Fully textured – Maximum grip for chemical handling
- Smooth surface – Lowest particle generation for ultra-clean environments
Wet conditions can affect tactile sensitivity, so texture choice impacts both safety and contamination control.
Top Manufacturers and Innovations
Leading manufacturers have invested heavily in pharmaceutical-grade nitrile gloves:
Hartalega – Pioneered ultra-thin nitrile technology with enhanced tactile sensitivity while maintaining barrier protection.
Ansell – Developed specialized cleanroom gloves with validated low particle counts and chemical resistance data.
Kimberly-Clark – Offers nitrile gloves specifically tested for pharmaceutical cleanroom compliance with detailed particle generation reports.
Top Glove & Kossan – Major suppliers focusing on powder-free formulations meeting international GMP standards.
Recent innovations include:
- Polymer blends that reduce particle shedding by 40% compared to standard nitrile
- Antimicrobial coatings that inhibit bacterial growth on glove surfaces
- Color-coding systems to prevent cross-contamination between different manufacturing zones
- Extended cuff lengths (12+ inches) for better coverage under cleanroom gowns
Best Practices for Using Nitrile Gloves in Pharmaceutical Settings
Even the highest-quality gloves fail if used improperly. Nitrile Gloves in Pharmaceuticals: Compliance with GMP Standards and Particle Control depends on rigorous protocols.
Proper Donning Procedures
The sterile gloving technique is critical in pharmaceutical cleanrooms:
- Hand hygiene – Wash hands thoroughly with pharmaceutical-grade soap and dry completely
- Inspection – Check gloves for tears, holes, or contamination before donning
- Aseptic technique – Touch only the inside cuff when pulling on the first glove
- Cuff coverage – Ensure glove cuffs extend over gown sleeves by at least 2 inches
- Double-gloving – For sterile operations, don the second pair using sterile technique
Many facilities use best practices for protecting hands safely as part of their standard operating procedures.
Change Frequency and Contamination Prevention
GMP standards require glove changes at specific intervals:
- Every 30-60 minutes during continuous aseptic operations
- Immediately after touching non-sterile surfaces
- Between different products to prevent cross-contamination
- After any breach (tear, puncture, chemical exposure)
- When entering/exiting critical manufacturing zones
Cross-contamination risks 🔴 include:
- Transferring active pharmaceutical ingredients (APIs) between products
- Introducing environmental contaminants into sterile zones
- Spreading microbial contamination from non-sterile areas
- Chemical residue transfer affecting product quality
Storage and Handling
Proper storage maintains glove integrity:
- Temperature control – Store at 15-25°C (59-77°F) away from heat sources
- Humidity management – Maintain 30-70% relative humidity to prevent material degradation
- UV protection – Keep gloves away from direct sunlight and UV exposure
- Original packaging – Keep gloves sealed until use to minimize particle exposure
- First-in, first-out – Rotate stock to use gloves before expiration dates
Understanding the role of sulfur in nitrile gloves helps explain why proper storage matters—chemical accelerators can degrade over time if exposed to improper conditions.
Addressing Common Challenges and Concerns
Allergic Reactions and Sensitivities
While nitrile is latex-free, some individuals experience reactions to chemical accelerators used in manufacturing. Allergic reactions to nitrile gloves are rare but possible, typically presenting as contact dermatitis.
Solutions include:
- Accelerator-free nitrile formulations
- Hypoallergenic glove liners
- Regular hand moisturizing to prevent skin breakdown
- Rotation between different glove brands to identify triggers
Cost Considerations vs. Compliance
Pharmaceutical-grade nitrile gloves cost 2-3 times more than standard disposable gloves. However, the cost of non-compliance far exceeds glove expenses:
- Batch rejection – A single contaminated batch can cost $500,000-$5 million
- Regulatory fines – FDA warning letters and consent decrees carry substantial penalties
- Reputation damage – Product recalls destroy consumer trust and market value
- Legal liability – Contaminated products causing patient harm result in massive lawsuits
The nitrile gloves market is projected to reach USD 13.70 billion by 2036, reflecting the pharmaceutical industry’s commitment to quality over cost-cutting.
Environmental and Sustainability Concerns
Disposable gloves create significant waste. Whether nitrile gloves can be recycled remains a challenge, though some programs now exist for post-use collection and processing.
Sustainable practices include:
- Right-sizing glove usage to avoid unnecessary changes
- Implementing glove recycling programs where available
- Choosing manufacturers with environmental certifications
- Exploring biodegradable nitrile formulations (still in development for pharmaceutical use)
The Future of Nitrile Gloves in Pharmaceutical Manufacturing
The pharmaceutical industry continues evolving, and glove technology advances alongside it. Companies like DuPont and Kimberly-Clark are developing next-generation nitrile gloves optimized for ISO Class 1-5 cleanrooms with even lower particle generation and enhanced chemical resistance.
Emerging trends include:
- Smart gloves with embedded sensors detecting breaches or contamination
- Antimicrobial formulations that actively kill pathogens on contact
- Improved sustainability through bio-based nitrile alternatives
- Enhanced tactile sensitivity rivaling bare-hand dexterity
- Extended wear formulations reducing change frequency without compromising safety
The medical nitrile gloves market is expected to reach USD 14.88 billion by 2034, with pharmaceutical applications driving much of this growth as vaccine development, biologics manufacturing, and personalized medicine expand.
Conclusion
Nitrile Gloves in Pharmaceuticals: Compliance with GMP Standards and Particle Control represents a critical intersection of worker safety, product quality, and regulatory compliance. As pharmaceutical manufacturing becomes increasingly sophisticated, the humble nitrile glove has evolved into a highly engineered barrier against contamination.
Powder-free, low-particulate nitrile gloves are no longer optional—they’re mandatory for GMP compliance. With the cleanroom gloves market growing at 4.7% annually and regulatory standards tightening, pharmaceutical facilities must prioritize proper glove selection, rigorous usage protocols, and continuous staff training.
Take Action Now ✅
- Audit your current glove inventory – Ensure all gloves meet ISO cleanroom classifications for your operations
- Review change protocols – Update SOPs to reflect current GMP best practices
- Train staff thoroughly – Proper donning and contamination prevention techniques are essential
- Partner with reputable suppliers – Choose manufacturers providing particle count validation and GMP compliance documentation
- Monitor regulatory updates – Stay current with FDA, EMA, and ISO standard revisions
The pharmaceutical industry’s commitment to quality means that every detail matters—including the gloves protecting both workers and products. By understanding and implementing proper Nitrile Gloves in Pharmaceuticals: Compliance with GMP Standards and Particle Control practices, facilities can maintain the highest standards of drug manufacturing while safeguarding patient health worldwide.
SEO Meta Title: Nitrile Gloves in Pharma: GMP Compliance & Particle Control