Healthcare workers in India change gloves dozens of times per shift, exposing their hands to chemical accelerators that trigger allergic contact dermatitis in up to 10% of frequent users. In March 2026, Wadi Surgicals launched Enliva—India’s first accelerator-free nitrile gloves—eliminating the thiurams, carbamates, and MBTs that cause Type IV hypersensitivity reactions. This breakthrough brings world-class skin-friendly protection to Indian hospitals, labs, and food facilities at accessible prices, matching global standards set by brands like Ansell and Hartalega.

Key Takeaways
✅ Enliva eliminates chemical accelerators (thiurams, carbamates, MBTs) that cause allergic contact dermatitis in healthcare workers
✅ EN 455 and ISO 374 certified for medical use, chemical resistance, and mechanical durability
✅ Produced in Hyderabad at 1 million gloves daily (300 million annually) with Rs 100 crore expansion planned
✅ Affordable alternative to imported hypoallergenic gloves, making skin-safe protection accessible across India
✅ Suitable for multiple sectors: healthcare, biotech labs, food processing, electronics, and cleanrooms
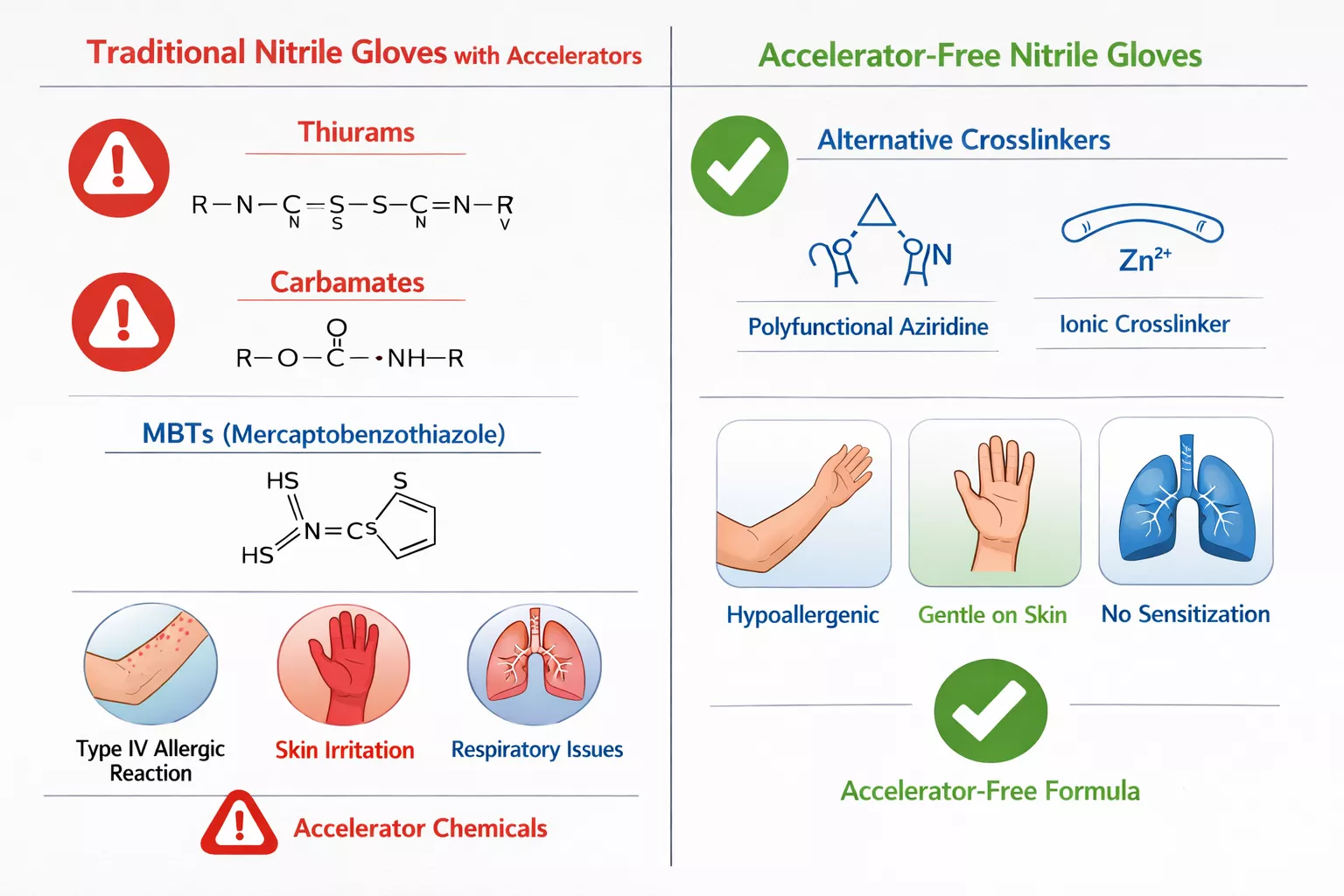
What Makes Accelerator-Free Nitrile Gloves Different?
Traditional nitrile gloves rely on chemical accelerators during manufacturing to speed up the vulcanization process—the chemical reaction that gives gloves their elasticity and strength. Common accelerators include:
- Thiurams (tetramethylthiuram disulfide)
- Carbamates (zinc diethyldithiocarbamate)
- MBTs (mercaptobenzothiazole)
These chemicals remain as residues in finished gloves and can leach onto skin during prolonged wear. For healthcare workers who don and doff gloves 50+ times daily, this repeated exposure triggers Type IV hypersensitivity—a delayed allergic reaction causing redness, itching, blistering, and painful dermatitis.
Understanding allergic reactions to nitrile gloves is crucial for anyone working in medical settings. The symptoms can range from mild irritation to severe contact dermatitis that forces workers to leave their professions.
The Enliva Solution 🧤
Enliva uses alternative non-sensitizing crosslinking agents instead of traditional accelerators. These proprietary compounds achieve the same mechanical properties—flexibility, tensile strength, chemical resistance—without leaving allergenic residues. Wadi Surgicals collaborated with global polymer experts to develop this formulation, ensuring the gloves meet international benchmarks for:
- Durability: Withstand punctures and tears during medical procedures
- Flexibility: Maintain tactile sensitivity for delicate tasks
- Protection: Resist chemicals, bloodborne pathogens, and contaminants
The result? Gloves that feel identical to standard nitrile but dramatically reduce skin reactions. For more context on what nitrile gloves are made of, the base material remains acrylonitrile-butadiene rubber—only the curing process changes.
Enliva’s Certifications and Standards for Healthcare Safety
Accelerator-Free Nitrile Gloves: India’s Enliva Brand and Reducing Allergic Risks for Healthcare Workers isn’t just a marketing claim—Enliva backs it with rigorous testing and international certifications:
| Certification | What It Means | Why It Matters |
|---|---|---|
| EN 455 | European standard for medical gloves (freedom from holes, physical properties, biocompatibility) | Ensures gloves are safe for patient care and surgical use |
| EN ISO 374 | Chemical resistance and protection against microorganisms | Confirms barrier protection against bloodborne pathogens and hazardous chemicals |
| ISO 13485:2016 | Quality management for medical devices | Guarantees consistent manufacturing processes and product safety |
| REACH Compliance | EU regulation restricting hazardous substances | Verifies gloves are free from banned chemicals and safe for European markets |
These certifications position Enliva alongside global leaders like Ansell’s DermaShield, Hartalega’s NGX, and SHIELD Scientific. The gloves are suitable for:
- 🏥 Healthcare: Hospitals, clinics, dental offices, emergency services
- 🔬 Biotech Labs: Pharmaceutical research, microbiology, pathology
- 🍽️ Food Processing: Kitchens, packaging facilities, quality control
- 💻 Electronics: Cleanrooms, semiconductor manufacturing, precision assembly
- 🧪 Industrial: Chemical handling, automotive, janitorial services
For workers concerned about nitrile allergic reactions, Enliva’s dermatological testing confirms significantly lower sensitization rates compared to accelerator-containing gloves.

How Enliva Reduces Allergic Risks for Healthcare Workers
The Allergy Problem in Healthcare 🩺
Healthcare workers face a triple threat when it comes to glove-related skin issues:
- Frequent glove changes: Nurses and doctors may use 50-100 gloves per shift
- Prolonged wear: Surgeons wear gloves for hours during procedures
- Hand hygiene protocols: Repeated handwashing and sanitizing strips skin’s protective barrier
When accelerator chemicals contact compromised skin, the immune system mounts a delayed response (Type IV hypersensitivity). Symptoms appear 24-72 hours after exposure and include:
- Redness and swelling on hands and wrists
- Itchy, dry, cracked skin
- Painful blisters and eczema-like rashes
- Thickened, scaly patches (chronic cases)
For detailed information on allergic reactions to nitrile gloves, the condition can become debilitating enough to require job modifications or career changes.
Enliva’s Impact on Worker Safety 💪
By removing accelerators, Enliva gloves eliminate the primary trigger for Type IV allergies. Anindith Reddy, Managing Director of Wadi Surgicals, emphasized that the brand addresses accessibility barriers—imported hypoallergenic gloves cost 2-3x more than standard nitrile, making them unaffordable for many Indian healthcare facilities.
Enliva’s competitive pricing (achieved through local manufacturing in Hyderabad) means:
- More facilities can afford skin-safe gloves for all staff, not just those with diagnosed allergies
- Preventive protection reduces new allergy cases among workers
- Improved retention as fewer healthcare professionals leave due to occupational dermatitis
Selvam Subramaniam, Vice President of Operations at Wadi Surgicals, credits advanced material science for maintaining performance while eliminating sensitizers. The gloves match international standards for tensile strength, elongation, and barrier protection—proving that safety and skin health aren’t trade-offs.
Enliva’s Production Scale and Future Plans
Current Manufacturing Capacity 🏭
Wadi Surgicals’ Hyderabad facility produces 1 million nitrile gloves daily—approximately 300 million gloves annually. This output serves:
- Domestic markets: Indian hospitals, clinics, pharmaceutical companies, food processors
- Export markets: Asia-Pacific, North America, and Europe (driven by REACH compliance)
The company was founded in 2021 during the COVID-19 pandemic, when global glove shortages highlighted India’s dependence on imports from Malaysia and Thailand. Enliva represents India’s push toward self-reliance in critical medical supplies.
Rs 100 Crore Expansion Investment 💰
In early 2026, Enliva announced a Rs 100 crore investment to scale operations, including:
- Deeper R&D: Developing sector-specific variants (e.g., extended cuff for surgical use, textured grip for wet environments)
- White-label and OEM options: Partnering with international distributors to expand global adoption
- Increased production: Targeting 2 million gloves daily by 2027
- Quality enhancements: Advanced testing labs for dermatological safety and chemical resistance
Anindith Reddy stated the investment will position Enliva as a global player in the medical disposables market, competing directly with established brands while maintaining affordability.
Sector-Specific Innovations 🔬
Enliva plans to launch specialized gloves for:
- Surgical suites: Longer cuffs, sterile packaging, enhanced tactile sensitivity
- Food safety: FDA compliance, textured grip for handling wet ingredients
- Cleanrooms: Low-particulate formulations for electronics and pharmaceutical manufacturing
- Industrial applications: Thicker gauges for chemical handling and automotive work
For those exploring the best thickness for nitrile gloves, Enliva’s upcoming range will offer 3-8 mil options tailored to specific hazards.

Why Accelerator-Free Nitrile Gloves Matter in 2026
Global Shift Toward Hypoallergenic PPE 🌍
The accelerator-free movement isn’t unique to India—it reflects a worldwide trend toward safer personal protective equipment. European hospitals increasingly mandate accelerator-free gloves under occupational health regulations, and U.S. healthcare systems prioritize them to reduce workers’ compensation claims for dermatitis.
Enliva’s launch aligns India with this global standard, ensuring Indian healthcare workers receive the same protections as their counterparts in developed nations. The brand’s REACH compliance and EN certifications facilitate exports, positioning India as a manufacturing hub for next-generation medical gloves.
Addressing the Latex Allergy Crisis 🚫
While nitrile gloves were originally developed as a latex-free alternative (avoiding Type I latex protein allergies), accelerator chemicals created a new allergy problem. Enliva solves both issues:
- No latex proteins: Safe for workers and patients with latex allergies
- No accelerator chemicals: Eliminates Type IV hypersensitivity risks
For comprehensive information on latex allergy types, understanding the distinction between protein allergies (Type I) and chemical allergies (Type IV) is essential for selecting appropriate gloves.
Economic and Social Impact 💼
Enliva’s affordable pricing democratizes access to premium gloves, particularly benefiting:
- Rural healthcare facilities with limited budgets
- Small clinics and dental practices that couldn’t afford imported hypoallergenic gloves
- Food service workers in restaurants and catering businesses
- Laboratory technicians in academic and research institutions
By reducing occupational dermatitis, Enliva also lowers indirect costs: sick leave, medical treatment, job retraining, and lost productivity. The social impact extends to worker well-being and career longevity in healthcare professions.
Comparing Enliva to Global Accelerator-Free Brands
| Brand | Origin | Key Features | Price Point |
|---|---|---|---|
| Enliva | India | EN 455/ISO 374 certified, REACH compliant, 300M annual production | Affordable (local manufacturing) |
| Ansell DermaShield | Malaysia | Polymer coating, low dermatitis potential, FDA approved | Premium (imported) |
| Hartalega NGX | Malaysia | Nitrile with softness additives, accelerator-free, eco-friendly | Premium (imported) |
| SHIELD Scientific | Malaysia | Accelerator-free, textured grip, medical-grade | Mid-to-premium (imported) |
Enliva’s competitive advantage lies in local production and price accessibility without compromising quality. The brand’s certifications match international standards, making it a viable substitute for imported gloves in most applications.
How to Choose the Right Accelerator-Free Gloves
When selecting gloves for your facility, consider:
- Application: Medical procedures require EN 455 certification; food handling needs FDA compliance
- Thickness: 3-4 mil for exams, 5-6 mil for lab work, 7-8 mil for industrial tasks
- Texture: Smooth for general use, textured for wet or oily environments
- Cuff length: Standard (9-10 inches) or extended (12 inches) for forearm protection
- Sterility: Sterile for surgical use, non-sterile for exams and general tasks
For guidance on sterile vs. non-sterile gloves, the choice depends on infection control requirements and procedure type.
Conclusion: A Safer Future for Healthcare Workers
Accelerator-Free Nitrile Gloves: India’s Enliva Brand and Reducing Allergic Risks for Healthcare Workers represents a milestone in occupational health. By eliminating thiurams, carbamates, and MBTs, Enliva protects the hands that protect us—nurses, doctors, lab technicians, and food handlers who rely on gloves for safety.
Wadi Surgicals’ March 2026 launch brings world-class hypoallergenic protection to India at accessible prices, backed by EN 455 and ISO 374 certifications. With 300 million gloves produced annually and a Rs 100 crore expansion underway, Enliva is poised to become a global leader in skin-safe PPE.
Take Action Today 🎯
- Healthcare facilities: Request Enliva samples to test with your staff
- Procurement managers: Compare costs against imported accelerator-free brands
- Workers with glove allergies: Ask your employer about switching to Enliva
- Distributors: Explore white-label and OEM partnership opportunities
The future of hand protection is accelerator-free—and India is leading the way. Protect your team’s skin health while maintaining the barrier protection that saves lives. Enliva proves that safety, comfort, and affordability can coexist in a single glove.